In plastic injection molding, the quality of the products is the cornerstone for enterprises to establish themselves in the market, and quality controllers play a crucial role in the quality control process. From pre-production preparations to shift handover, every step can affect the quality of the product. This article provides a comprehensive guide for quality controllers to help them understand their key responsibilities and carry out the quality control process efficiently. If you are in the position of quality controller, please make sure that you adhere strictly to the following rules.
- 1. An Overview of the Inspection Process
- 2. Pre-Production Preparation: Laying the Foundations of High-quality Products
- 3. First Article Inspection: Establishing Production Control
- 4. Coaching After First Article Inspection: Unifying the Standards
- 5. Inspection Record: Monitoring the Production Process
- 6. Shift Handover: Ensuring Greater Continuity of Work
- 7. Basic Management: Asset and Document Control
- Conclusion
1. An Overview of the Inspection Process
Pre-production preparation → First Article Inspection → In-process Inspection & Defect Confirmation → Pre-delivery Inspection → Identification of Non-conforming Products → Shift Handover
2. Pre-Production Preparation: Laying the Foundations of High-quality Products
Make the following preparations according to the Daily and Weekly Production Plan of Injection Molding to ensure that the inspection process starts in order:
- Prepare all relevant sealed samples: sealed samples of injection-molded parts, color chips, color samples, visual quality standards, and size standards.
- Collect a full set of technical documents: Injection Molding Part Drawing, Inspection Standards of Injection Molding Parts, Injection Molding Material List, Process Specification, Mold Modification Notice, and Design Change Notice.
- Prepare inspection tools and fixtures: vernier calipers, depth gauges, inside calipers, etc., and verify that their accuracy meets the required specifications.
- Prepare mating parts for subsequent assembly inspection.
3. First Article Inspection: Establishing Production Control

3.1 Timing
The first article inspection process must be carried out in the following situations: machine shutdown, mold changing, material changing, core replacement, process adjustment, and shift handover.
3.2 Basis
The following materials shall serve as the primary criteria for evaluation: the Injection Molding Part Drawing, Inspection Standards of Injection Molding Parts, Injection Molding Material List, sealed samples in the product design process, color chips, and color samples.
3.3 Content
3.3.1 When the process is stable, inspect the appearance (including color), function, structure, critical dimensions, and mating parts for all cavities of a full set of molded parts. Record the results in the Form of First Article Inspection in Injection Molding.
3.3.2 For parts that have mating parts, examine them with the corresponding mating parts.
3.3.3 If a measured dimension does not conform to the standard but matches the sealed sample, the sealed sample shall serve as the basis for judgment.
3.3.4 After the first article inspection is approved, notify the production department to start mass production. Record the quality control material for this batch of parts and place it in a specified location on the machine as a reference for operators.

3.4 Defect Report
3.4.1 If any quality or material defect is found during the first article inspection process, isolate and label it at once, then notify the team leader and production manager to solve the problem.
3.4.2 If the on-site team leader has adjusted the machine beyond 50 cycles and the problem is still not solved, notify the production manager immediately. If the problem is not solved within 1 hour and it affects the function of the product, the production process should be stopped;
3.4.3 For defects in quality and technique that cannot be immediately resolved by the team leader or production manager, issue a Defect Report of quality control material if necessary. After being signed by the general manager, it should be handed over to the Production Department or Quality Control Department. If the problem is so serious that the machine needs to be turned off, the team leader of injection molding or factory manager should be notified, and the team leader is required to sign the Form of First Article Inspection in Injection Molding.
3.4.4 After receiving the defect report, the responsible department should analyze the cause of the defect and propose measures for improvement, then send them to the unit which detected the problem and the relevant units for implementing the measures.
3.4.5 File the defect report for the quality control material, track the problem-solving process, and confirm that the problem is resolved.
4. Coaching After First Article Inspection: Unifying the Standards
Targeted coaching is required based on categories such as electroplated parts, welded parts, mating parts, and colors. The core content is as follows:
4.1 Operation Techniques
Key operating procedures for observation, handling, trimming range and degree, wiping position and method, etc. of the product must be clearly defined to ensure that operators all have a good command of them.
4.2 Quality Requirements for Classified Products
4.2.1 Exterior Parts
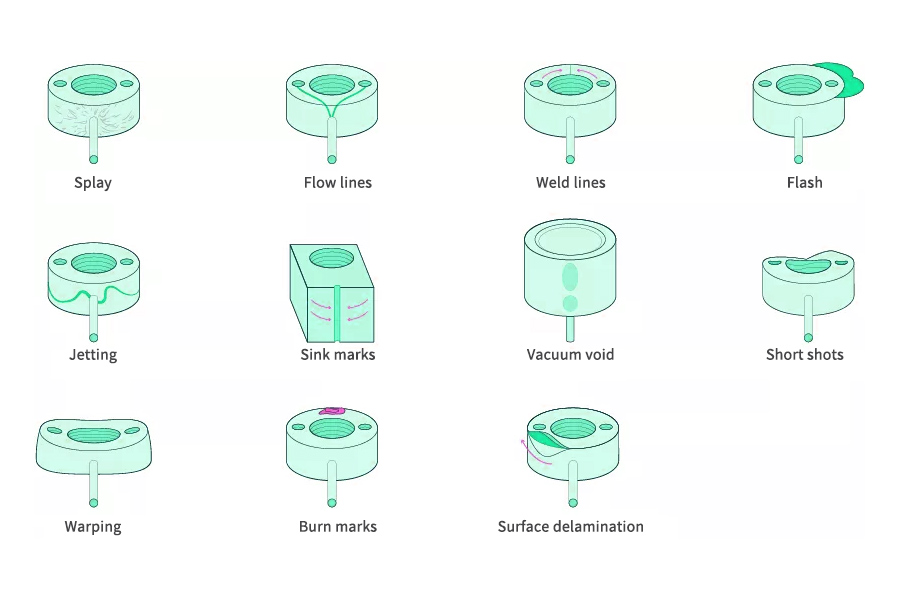
- Electroplated Products: Common defects include pitting, cold slug, splay, flow marks, sink marks, flow lines, weld lines, etc. Highly defective products should be selected, polished, or scrapped, while slightly defective products are acceptable and require on-site sampling for reference and judgment by operators. Electroplated parts with poor exterior need to be polished and marked.
- Compatibility Requirements: Products that need to be matched shall be inspected every hour, and if necessary, 100% actual matching shall be carried out. The actual matching standard shall refer to the first article.
4.2.2 Interior Parts
- Sealing Surface: There should be no scratches, mold strains, sink marks, obvious weld lines, or other concave and convex defects on the sealing surface, and the dimensions should be within the tolerance range.
- Welding Surface: There should be no obvious sink marks, scratches, strains, splay, ejector pin marks, or raised ejector pins on the welding surface, and there should be no cracks on any welding rib.
- Compatibility: Products that require matching shall undergo compatibility inspections every hour, and 100% inspection shall be conducted if necessary.
4.3 Special Precautions
4.3.1 When producing various types of fast-food containers, it is necessary to check whether the products are prone to cracking, whether the wall thickness around them is uniform, whether they can be hermetically sealed with their mating parts, and what problems have occurred previously or may occur.
4.3.2 Products with high quality requirements are classified as 1st-level products, while products with normal requirements are classified as 2nd-level products. Make sure that the overall dimensions are consistent.

4.4 Packaging and Protection Specifications
4.4.1 Packaging Requirements: Refer to the List of Packaging Bases and ensure that all operators have a clear understanding of the correct packaging bases and methods.
4.4.2 Product Protection: Handle the products with care to avoid collision, scratches, and damage. When bagging, ensure that they are properly placed and not exposed. When packing, the products should not exceed the edge of the box.
4.5 Verification of Coaching Effectiveness
For specific coaching projects, you can also refer to the Quality Coaching for First Article Operators. After explaining all sorts of required operating methods, it is necessary to examine whether the operator understands what you have said by checking whether the products he or she has inspected meet the standards. If there is anything that the operator is unclear about, you must re-explain and demonstrate the procedure until he or she fully understands and is able to operate independently.
5. Inspection Record: Monitoring the Production Process

5.1 Basic Requirements for Inspection
The quality controller shall inspect the machine at least once every two hours, and products in no less than four molds shall be randomly selected each time. Please pay more attention to the following tasks:
- Product Inspection: Inspect the exterior (including color), dimensions, structure, function, and compatibility of the product based on the drawings, master samples, and part inspection standards, then record the results in the Inspection Record Form for Injection Molding.
- Operation Specification Inspection: Check whether the operators’ operation methods are correct, whether they have carried out mutual inspection, and whether the operation is standardized. If non-standard operations are found, handle them according to the degree of quality impact: notify the operator and supervisor for the first time; notify the operator, supervisor, and injection molding supervisor for the second time; notify the operator, quality control manager, and production manager for the third time.
- Process and Document Inspection: Confirm that the machine parameters are consistent with the process card, and notify the supervisor to make adjustments if they exceed the tolerance range. If the adjustment according to the process card cannot meet the requirements and the process needs to be changed, the team leader should be notified and the factory manager should be informed, and the process card should be updated synchronously. At the same time, check whether the indispensable documents such as the Process Card, Machine Inspection Form, Inspection Standards, Operation Manual, etc. are placed on the machine.
5.2 Judgement Criteria for Sampling Inspection
5.2.1 Among the qualified products self-inspected by the operator, if the number of functional and structural non-conforming products is more than 2 pieces, or the number of exterior non-conforming products is more than 3 pieces, then the products from the previous period can be judged as non-conforming.
5.2.2 Among the operator-selected or post-processed products, if the number of non-conforming products or scrap products is more than 3 pieces, then the products from the previous period can be judged as non-conforming.
5.2.3 After the inspection is judged as unqualified, the quality controller needs to record it in the Inspection Record Form for Injection Molding. The Inspection Record Form should be confirmed and signed by the supervisor before the products are isolated and reprocessed. After reprocessing, the products must be self-inspected by the operator before being sent for inspection.
5.2.4 For the full box products selected by the operator, stamp the quality control seal after passing the sampling inspection and confirm whether the information such as the part number, name, quantity, date, and special identification is correct. If the products fail the sampling inspection, a seal for reprocessing shall be affixed, and the team leader and operator shall be notified for reprocessing.
5.3 Defect Report and Handling
5.3.1 For defects that can be immediately remedied, record in detail the major defects, measures for improvement, and the result in the Inspection Record Form for Injection Molding.
5.3.2 For defects that cannot be immediately resolved or cross shifts, issue a Defect Report, submit the exterior defect report to the general manager for judgment, and submit the functional or structural defect report to the production department for processing after it is confirmed by the quality control department. Defect reports are distributed by the quality control department to relevant departments for implementation, and the quality controller is responsible for tracking the response and implementation of countermeasures.
5.4 Identification of Non-conforming Products
5.4.1 Before each inspection and shift handover by the quality controller, it is necessary to randomly check the scrapped boxes to confirm whether there are any good products mixed in with the defective ones and handle the problems promptly upon discovery.
5.4.2 Defective products with less than 20 molds can be scrapped after being confirmed by the injection molding factory and the quality controller. For molds exceeding 20 times, they must receive confirmation from the injection molding workshop, quality inspection, and production department and approval from the general manager before being scrapped. When necessary, the quality control department shall issue a Defect Report to the injection molding workshop, which shall be submitted to the injection molding workshop for cause analysis and the formulation of countermeasures after confirmation from the production department, and the quality controller is required to track the problem-resolving process.
6. Shift Handover: Ensuring Greater Continuity of Work
6.1 The quality controller of the next shift shall hold the Handover Record and inspect the condition of every machine with the quality controller of the previous shift. The products’ quality, first article, and other key information of each machine in the previous shift shall be examined separately, and every abnormal condition found shall be handed over clearly. If necessary, feedback shall be given to the production department according to the regulations.
6.2 Every defect that has occurred in this shift and how it has been dealt with should be recorded in detail in the Handover Record along with the work precautions, and the record must be fully handed over to the next shift.
6.3 All types of forms, documents, and other important notifications received on the same day must be recorded in the Handover Record and handed over to the next shift.
7. Basic Management: Asset and Document Control
7.1 Properly archive documents and records; strengthen the control of samples, testing fixtures, and quality control seals; and regularly maintain and safely store them to ensure that all resources are traceable and in good condition.
7.2 The closed-loop process of handling defects in quality
7.2.1 The OK Process: The Quality Control Department issues the notification. → The Injection Molding Workshop distributes the notification. → The Responsible Department implements measures for improvement. → The Quality Control Department tracks the results. → The General Manager confirms their effectiveness.
7.2.2 The NG Process: The quality control department issues the notification. → The Injection Molding Workshop hands it over to the Production Department. → The Quality Control Department distributes the notification. → The Responsible Department implements measures for improvement. → The Quality Control Department tracks the results. → The General Manager confirms their effectiveness.
Conclusion
Quality control is no trivial matter, and every rigorous and meticulous step lays the foundation for the high quality of products. We sincerely hope that all quality controllers will strictly adhere to this guide when carrying out their work, proactively identify and solve problems in a timely manner, and jointly build the quality defense line of the enterprise. If the content of the guide needs to be adjusted based on actual producing conditions, you are welcome to give us feedback for optimization at any time.
For quality controllers, the toughest challenges often stem from the perennial problems that repeatedly occur and are difficult to cope with through on-site adjustments. As a leading injection molding company in China, KingStar is well versed in this field, and we focus on starting from the root of mold design and process to provide you with effective solutions. If you want to improve your products’ quality and enhance product competitiveness, KingStar is the right choice. Just feel free to contact us by email (sales@kingstarmold.com) or fill out a form on our contact page.



